 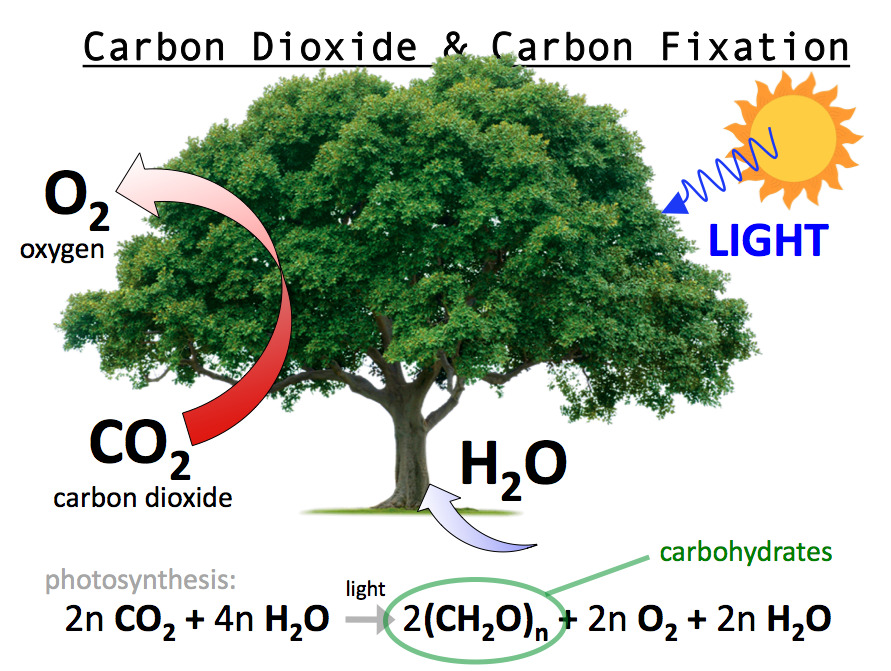
Detailed understanding of the principles behind pyrenoid assembly, regulation and structural organization within diverse lineages will provide a fundamental understanding of this biogeochemically important organelle and help guide ongoing efforts to engineer pyrenoids into crops to increase photosynthetic performance and yields. In this review, we summarise current knowledge about pyrenoid assembly, regulation and structural organization in Chlamydomonas and highlight evidence that LLPS is the general principle underlying pyrenoid formation across algal lineages and hornworts. Even though LLPS may underlie the apparent convergent evolution of pyrenoids, our current molecular understanding of pyrenoid formation comes from a single example, the model alga Chlamydomonas reinhardtii. Pyrenoids are the heart of algal and hornwort biophysical CO 2 concentrating mechanisms, which accelerate photosynthesis and mediate about 30% of global carbon fixation. Pyrenoids are formed by liquid-liquid phase separation (LLPS) of Rubisco, the primary CO 2 fixing enzyme, with an intrinsically disordered multivalent Rubisco-binding protein. The first half of the cycle, from acetyl-CoA to succinate-CoA, corresponds to the reductive citric acid cycle and the latter half of the cycle, from succinate-CoA to two molecules of acetyl-CoA, corresponds to the hydroxypropionate-hydroxybutyrate cycle.Pyrenoids are non-membrane bound organelles found in chloroplasts of algae and hornwort plants that can be seen by light-microscopy. Recent genome study suggests that this cycle may exist in Desulfurococcales (to which Ignicoccus belongs) and Thermoproteales (a taxon close to the origin of archaea). This cycle has been found only in Ignicoccus hospitals, a strictly anaerobic hyperthermophilic archaea. (v) The dicarboxylate-hydroxybutyrate cycle was named after its intermediates: succinate (a kind of dicarboxylate) and hydroxybutyrate. One complete turn of this cycle generates two molecules of acetyl-CoA, one of which is reutilized in the the cycle and the other is removed for cell material biosynthesis. Some of the intermediates and the carboxylation reactions are the same as in the 3-hydroxypropionate bicycle. (iv) The hydroxypropionate-hydroxybutyrate cycle is found in aerobic Crenarchaeota, Acidianus, Metallosphaera, and Sulfolobales. In addition, this bicycle provides the secondary benefit of useful intermediates for biosynthesis: acetyl-CoA, glyoxylate, and succinyl-CoA. In one complete turn of this bicycle, three molecules of bicarbonate are converted into one molecule of pyruvate. (iii) The 3-hydroxypropionate bicycle is found in some green non-sulphur bacteria of the family Chloroflexaceae. A bifunctional enzyme, carbon monoxide dehydrogenase/acetyl-CoA synthase, catalyzes the reactions from CO2 to CO and from CO2 to a methyl group, and then to generate acetyl-CoA. (ii) The reductive acetyl-CoA pathway (Wood-Ljungdahl pathway) is found in strictly anaerobic bacteria and archaea (Proteobacteria, Planctomycetes, Spirochaetes, and Euryarchaeota), some of which are methane-forming. In one complete turn of this cycle, four molecules of CO2 are fixed by the enzymes that are sensitive to oxygen, resulting in the production of one molecule of oxaloacetate, which is itself an intermediate of the cycle. (i) The reductive citric acid cycle (Arnon-Buchanan cycle) is found in microaerophiles and anaerobes, such as green sulfur bacteria. There are, at least, five additional carbon fixation pathways known to exist in autotrophic bacteria and archaea, which differ in reducing compounds, energy source, and oxygen sensitivity of enzymes. Plants and cyanobacteria fix CO2 as organic compounds using solar energy mainly by the reductive pentose phosphate cycle (also called Calvin cycle, Calvin-Benson cycle, or Calvin-Benson-Bassham cycle). Carbon fixation is an important pathway for autotrophs living in various environments. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
